Probiotic strains detect and suppress cholera in mice. Engineered lactobacilli display anti-biofilm and growth suppressing activities against Pseudomonas aeruginosa. Treatment of murine colitis by Lactococcus lactis secreting interleukin-10. CRISPR interference for rapid knockdown of essential cell cycle genes in Lactobacillus plantarum. Development of a RecE/T-assisted CRISPR–Cas9 toolbox for Lactobacillus. Portable CRISPR-Cas9(N) system for flexible genome engineering in Lactobacillus acidophilus, Lactobacillus gasseri, and Lactobacillus paracasei. CRISPR/Cas9-assisted seamless genome editing in Lactobacillus plantarum and its application in N-acetylglucosamine production. Genome editing using the endogenous type I CRISPR–Cas system in Lactobacillus crispatus. Lactococcus lactis as a live vector: heterologous protein production and DNA delivery systems. coli Nissle for the treatment of phenylketonuria: a first-in-human phase 1/2a study. 
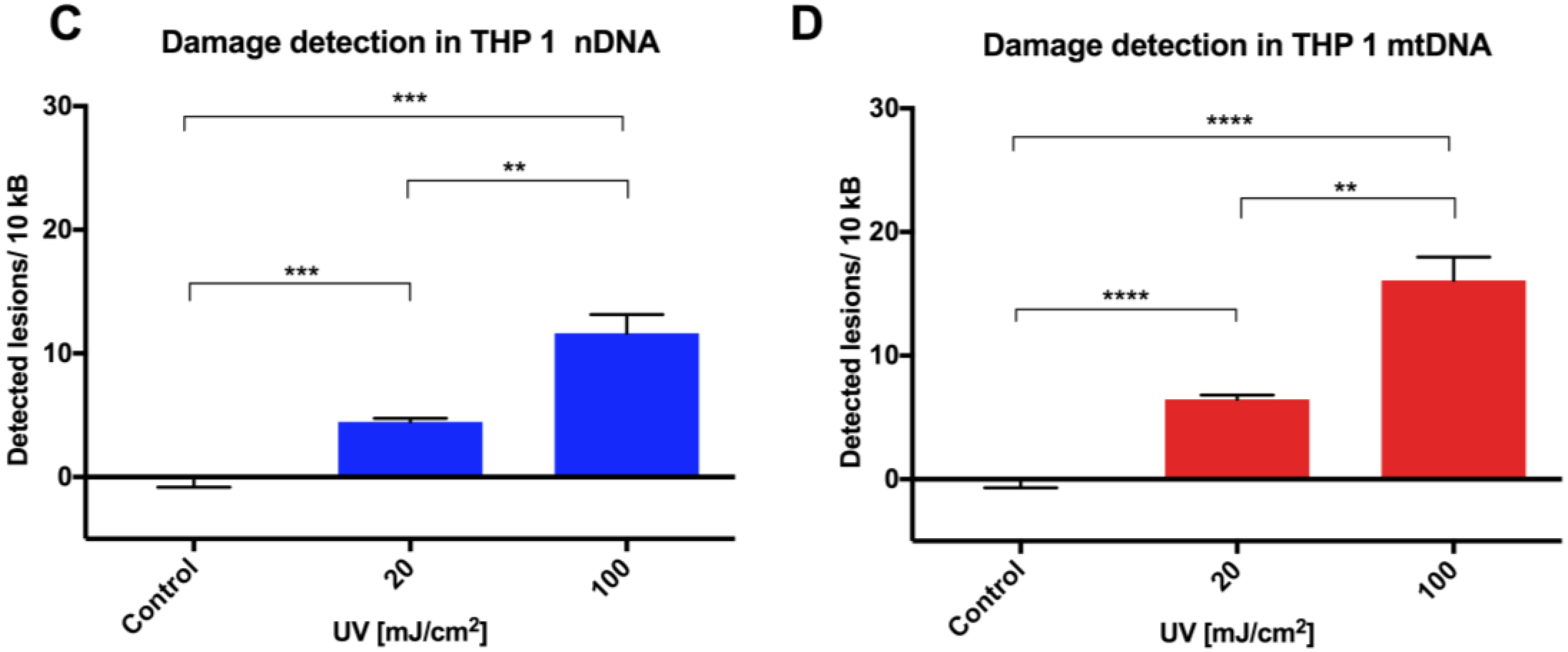
Safety and pharmacodynamics of an engineered E. Improvement of a synthetic live bacterial therapeutic for phenylketonuria with biosensor-enabled enzyme engineering. Engineered Escherichia coli Nissle 1917 with urate oxidase and an oxygen-recycling system for hyperuricemia treatment. coli Nissle improves hyperammonemia and survival in mice and shows dose-dependent exposure in healthy humans. Development of a synthetic live bacterial therapeutic for the human metabolic disease phenylketonuria. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Guild-based analysis for understanding gut microbiome in human health and diseases. Engineered bacteria as therapeutic agents. The gut microbiota - masters of host development and physiology. Structure, function and diversity of the healthy human microbiome. Massive expansion of human gut bacteriophage diversity. F., Almeida, A., Rangel-Pineros, G., Finn, R. Emerging technologies for gut microbiome research. Brain–gut–microbe communication in health and disease. Microbes in gastrointestinal health and disease. Understanding the holobiont: how microbial metabolites affect human health and shape the immune system. Urbanization and the gut microbiota in health and inflammatory bowel disease. This article reviews the role of the gut microbiome in human health and disease. The human intestinal microbiome in health and disease. Gut microbiota density influences host physiology and is shaped by host and microbial factors. 10.1038/s41466-5Ĭontijoch EJ, Britton GJ, Yang C, Mogno I, Li Z, Ng R, et al. Establishing microbial composition measurement standards with reference frames. Morton JT, Marotz C, Washburne A, Silverman J, Zaramela LS, Edlund A, et al. Compositional data analysis of the microbiome: fundamentals, tools, and challenges. Microbiome Datasets Are Compositional: And This Is Not Optional. Gloor GB, Macklaim JM, Pawlowsky-Glahn V, Egozcue JJ. Best practices for analysing microbiomes. Knight R, Vrbanac A, Taylor BC, Aksenov A, Callewaert C, Debelius J, et al. (c) and (d) show the associations between the qPCR-determined abundance of the butyryl-CoA:acetate CoA-transferase gene and the (c) estimated absolute abundance and (d) relative abundance of butyrate producers detected in the NGS data. The applied library preparation method (dual index TruSeq-tailed 1-step amplification ) causes a slight underestimation of Bacteroidetes abundance (unpublished data), explaining the underestimation observed for this phylum compared to qPCR. The correspondence decreases at the very low end of the abundance range, likely due to the relatively lower PCR amplification efficiency and increased stochasticity of the results for low abundance taxa in NGS. The dashed line shows the expected 1:1 correspondence. (b) Correlation between the qPCR abundances (16S rRNA gene copies per g feces) and the estimated absolute abundances of four taxa representing species, genus, family and phylum levels. qPCR-based estimate of copies of 16S gene per 1 g of feces. The top panel shows relative abundances based on 16S amplicon sequencing and the lower panels shows the estimated absolute abundances calculated by multiplying the relative abundances with total bacterial load, i.e. (a) Comparison of relative abundances and estimated absolute abundances of dominant bacterial families in 114 fecal samples. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
